50+
Hours saved per claims matrix
Emporia transforms clinical and economic literature into compliant, evidence-linked product claims so teams can move faster without sacrificing regulatory confidence.
Emporia helps teams compress query time, accelerate competition analysis, and reduce hours spent on matrix preparation before cross-functional review.
Hours saved per claims matrix
Annual savings across commercial teams
More relevant outputs than baseline models
Pilot in weeks, not quarters
Book a focused demo and we will walk through your exact workflow: evidence discovery, relevance ranking, and claims matrix generation with citation traceability.
Feature showcase
Toggle through claims matrix generation, AI literature search, and collateral generation without breaking the flow. Emporia keeps evidence, outputs, and review in one connected system.
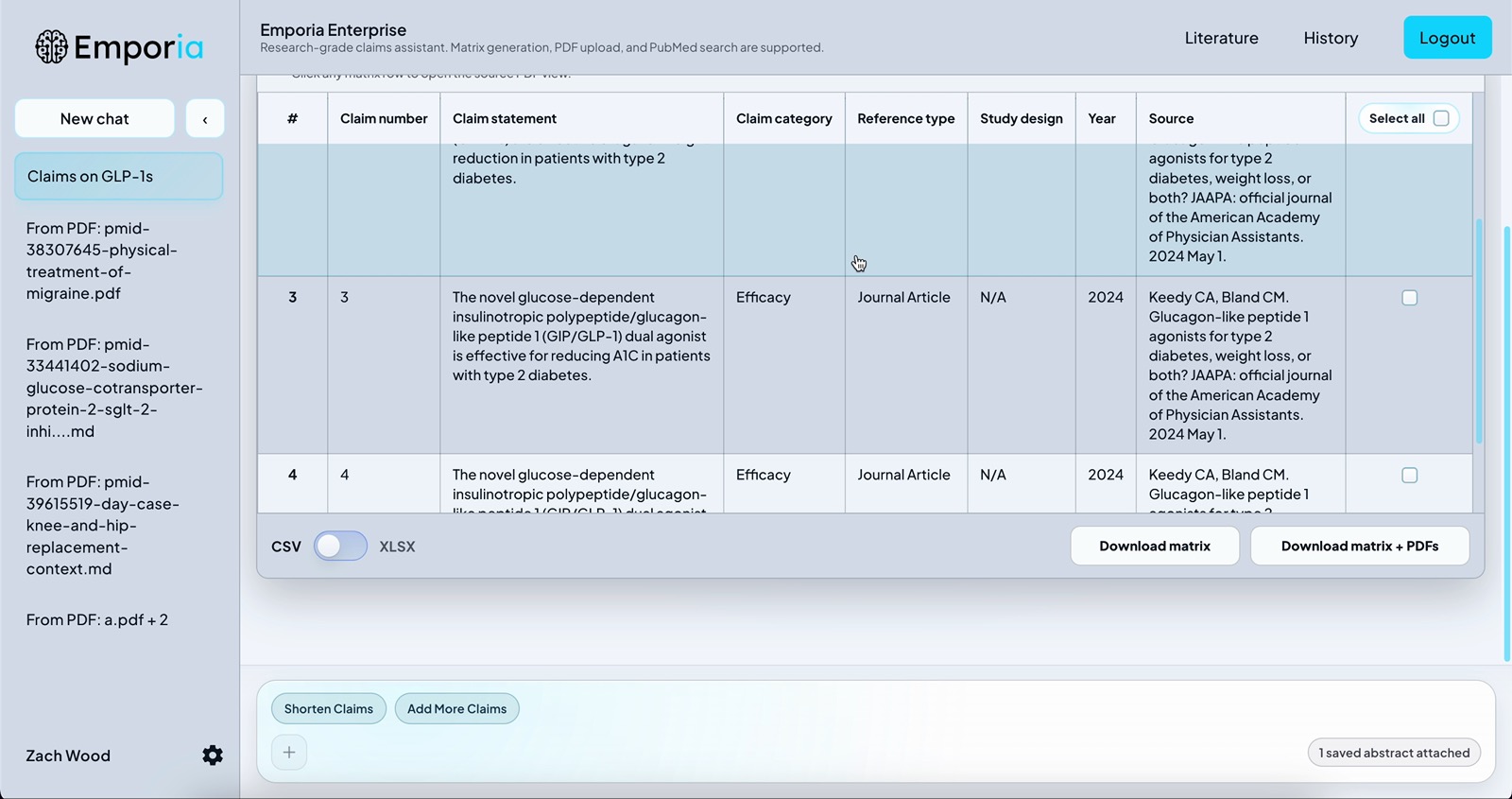
Claims matrix generation
Upload clinical studies, economic analyses, and regulatory documentation. Emporia extracts evidence across multiple PDFs and helps teams move toward a competitive matrix in minutes instead of days.
Key capability
Upload up to 10 PDFs (clinical, economic, or technical) in a single workspace, or attach saved abstracts from literature search. Emporia validates and prepares each source so claims are generated from the exact evidence you provide.
Key capability
Emporia extracts and structures product claims across your uploaded sources into a unified matrix complete with claim categories, study design, year, source, top authors, and citations.
Key capability
After matrix generation, Emporia produces annotated PDFs that highlight the exact text supporting each claim and labels it with the claim number, creating an audit-ready trail for medical, legal, and regulatory review.
How it works
Start with direct PDF uploads or run PubMed search and attach saved studies. Both paths converge into the same matrix generation and evidence-tracking export flow.
Start from your existing sources or discover new studies inside Emporia.
Bring selected sources into one chat before generation starts.
Use the same output flow regardless of where evidence came from.
Data handling and privacy
Emporia handles clinical and regulatory content with a controls-first approach across data protection, tenant isolation, model governance, and operational visibility.
Our team
We build Emporia at the intersection of life sciences and applied AI.
FAQs
Emporia currently supports two source paths: direct PDF uploads and saved PubMed literature items (including synthetic PDFs when full text is unavailable). You can combine these in one chat, with a current cap of 10 total sources.
Emporia generates a structured claims matrix from your source set, then runs claim tracking so teams can review annotated source PDFs alongside the matrix.
Each claim is linked back to source evidence through citations and claim-tracking annotations. Teams can open claims and verify supporting passages directly in the source documents.
Yes. You can upload PDFs, attach saved literature items, and generate one matrix from the combined set. In the current product, source setup must be finalized before the first generation turn, after which the source set is locked.
No. Emporia accelerates drafting and evidence organization, but final approval should always come from your internal medical, legal, and regulatory stakeholders. Emporia is built to make that review faster and more consistent, not to replace it.
Emporia aims to produce high-quality drafts grounded in your source documents, but outputs should be validated by your team, especially for nuanced clinical language. The workflow is designed to make verification easy by keeping claims tied to the underlying sources.
Emporia uses TLS in transit, Azure-managed encryption at rest, JWT-based authentication, and user-scoped data isolation patterns across storage layers. Customer content is processed in inference-only mode and is not used to train foundation models.
Yes. Emporia supports matrix exports in CSV and XLSX so teams can share outputs in existing review workflows.
Most teams can generate an initial claims matrix quickly and then iterate through review cycles with significantly less manual extraction work. The biggest wins show up when multiple stakeholders need to verify claims against the same set of sources.
Emporia is built to support life sciences teams of many sizes, from resource-constrained biotech groups to business-unit teams inside large organizations. We typically start with a focused pilot and expand based on results.
Yes, schedule a demo and we can walk through Emporia using your document set. We will also discuss your review workflow so outputs align with how your team approves and deploys claims.
Contact